Behind the scenes of drug development in pharmaceuticals and medicine are many professionals who labor so that clinical trials not only get done safely, successfully, and on time, but according to all regulations. One such professional is the Clinical Project Manager (CPM), the captain of the ship who navigates the research crew down the right route and all regulations followed.
What Is a Clinical Project Manager?
It is a person who plans, coordinates, and leads clinical trials. Clinical trials are trials that are performed to find out if new drugs, devices, or treatments are effective and safe in human patients.
The CPM oversees the entire trial. They make sure that:
- The trial is being performed to very high levels of medicine and law.
- Patients are protected.
- Physicians, researchers, and sponsors communicate.
- The trial is on time and within budget.
In short, they are the co-ordinator who makes sure of the whole process. They monitor it from start to finish without a hitch.
Why Is the Role Important?

Clinical trials are extremely complicated. They can take years to months and hundreds of patients, a couple of countries, and millions of dollars. Trials can get delayed, botched, or sabotaged on patient safety unless handled by a professional Clinical Project Manager.
The CPM ensures:
- Safety: Trial patients are not harmed.
- Compliance: All as per regulatory agencies (e.g., FDA in the USA).
- Efficiency: Trial is completed on time and money is not wasted.
- Communication: All are told and communicated with.
Key Responsibilities of a Clinical Project Manager
CPM work is enormous and consists of many activities. Some of the most important activities are:
Project Planning
- Making the trial schedule.
- Plan and spend money.
- Announcing resources, i.e., people and machinery.
Team Management
- Managing doctors, researchers, nurses, and data staff.
- Ensuring that all one in the group knows his/her job.
- Resolving conflicts between team members.
Regulatory Affairs
- Ensuring the trial is conducted according to Good Clinical Practice (GCP).
- Preparing government ministry reports.
- Auditing and inspection.
Monitoring the Trial
- Monitoring patient recruitment (recruiting volunteers).
- Monitoring progress against targets.
- Reading reports and conclusions on a regular basis.
Budget and Finance
- Keeping costs within budget.
- Negotiating with suppliers and vendors.
- Preparing financial reports for sponsors.
Reporting and Communication
- Producing progress reports to stakeholders.
- Reporting to sponsors and senior management.
- Providing reports of changes and results to the team.
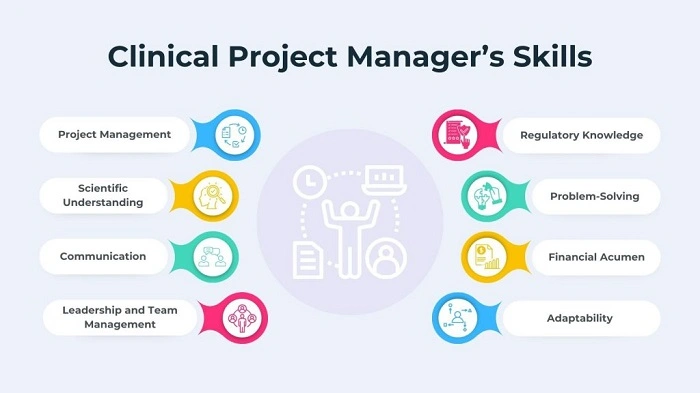
Skills to be a Clinical Project Manager
Technical skills, leadership skills, and communication skills should be possessed by a Clinical Project Manager in order to operate effectively.
Some of the most appropriate skills include:
- Organization: Managing many different tasks simultaneously.
- Leadership: Leadership and motivating an international team.
- Communication: Describing complex ideas to physicians, sponsors, and patients in simple language.
- Problem-Solving: Effective problem-solving in case a problem occurs.
- Attention to Detail: Ensuring any mistakes not related to patient safety or data.
- Time Management: Ensuring the project timeline.
- Regulatory Knowledge: Managing FDA, EMA, and global clinical trial regulation.
Career Path for Clinical Project Managers
Clinical Project Managers start their career mostly as Clinical Research Associates (CRAs) or in a similar role within research. They then move into project management after spending many years working and training.
The career progression typically occurs in the following sequence:
- Clinical Research Associate (CRA): First-level role responsible for managing sites clinically.
- Senior CRA / Clinical Trial Coordinator: Increased responsibility for sites and data.
- Clinical Project Manager (CPM): Overseeing total trials.
- Senior CPM or Director of Clinical Operations: Overseeing multiple projects.
- Executive Roles (VP, Head of Clinical Development): Total leadership.
Education and Training
Most progress to a Clinical Project Manager with:
- A bachelor’s degree in life science, pharmacy, nursing, or any other related degree.
- Some have higher degrees like a master’s or PhD.
- Clinical research (like ACRP or SOCRA) or project management (like PMP) certification is a plus.
Practical experience is significant. Recruiters like individuals who have gained experience in clinical research before turning their focus to management.
Issues for Clinical Project Managers
The work is rewarding but demanding. Some of the issues CPMs face are:
- Delays in Patient Recruitment: Struggling to get sufficient patients for trials.
- Budget Overruns: Over-spending occurs.
- Regulatory Pressure: Controlled by the government with strict regulations.
- Global Coordination: Trials need to be coordinated between nations whose legislations are different.
- High Stress: Patient safety and project success is in your hands.
Also Read: Sean Simpson: His Life, Career, and Legacy
Why This Role Is Rewarding
Despite the challenges, being a Clinical Project Manager is rewarding because:
- You actually contribute to the delivery of new drugs to patients.
- The job is diverse — each trial is unique.
- You have teams and become a leader.
- The job offers career progression and decent pay.
Remuneration and Career Opportunities
The United States salary for this position ranges from $90,000 to $130,000 annually. It depends on location and experience. Experienced Senior CPMs or those with large multinational companies can do even better.
The future career prospects look good. With the speedy demand for new treatments and medicines, clinical trials become more and more sought after, and this will translate into better job opportunities for CPMs.
Tips to be a Good Clinical Project Manager
If you are interested in pursuing this career, then read the below tips:
- Get some experience as CRA or clinical coordinator initially.
- Gain project management skills (i.e., Agile or PMP).
- Gain good communication and leadership skills.
- Stay abreast of clinical trial regulatory procedures.
- Contact industry experts.
FAQs
Is a Clinical Project Manager identical to a Clinical Research Associate?
No. A CRA is present at trial sites, whereas a CPM is at the project level.
Do Clinical Project Managers work in hospitals?
They will be working with pharmaceutical firms, research organizations, or biotech firms but will be spending tons of time in hospitals where trials are happening.
How stressful is the work?
It can sometimes be difficult with tight timelines, budgets, and keeping safety at the forefront, but it is an enjoyable job.
Can you work as Clinical Project Manager from home?
Yes. Most CPMs work from home, especially when they are working on international studies.
Do you require a medical degree?
No, but a health or life science background is extremely beneficial.
Conclusion
A Clinical Project Manager is not merely an official in charge of clinical trials, but an intermediary medicine, patients, and science. They bargain for themselves compliance, cost, safety, and schedule as they seek to bring medicines to market successfully and safely.
For those who are interested in science and desire to be leaders in teams and would like to serve with a job that can contribute to human lives, it is more suitable and worth becoming a Clinical Project Manager.